
Quantumbiochemistry.org
The quantum theory applied to biology
The time independent energy and wave-function for a molecule
In classical mechanics we describe a model as:
- a trajectory (also called an orbit) for the comprising particles
- the energy of the comprising particles.
In quantum mechanics the wave-function is the logical analogue for the trajectory.
We consider models that consist of moving electrons and atomic nuclei (a gas of electrons and atomic nuclei). Any molecule can thus be modeled as a gas of electrons and atomic nuclei and assume the total energy is the sum of :
- the kinetic energy of the electrons,
- the kinetic energy of the nuclei,
- the potential energy due the electrostatic forces between the nuclei,
- the potential energy due to the electrostatic forces between the electrons,
- the potential energy due to the electrostatic forces between the electrons and the nuclei.
With this in mind we can write down a general expression for the time independent Schrödinger equation for molecules.
- ψ is the unknown molecular wave-function
- E the unknown energy and
- H the molecular Hamiltonian.
The hamiltonian H for a model with i(1 → N) electrons and A (1 → M ) nulceï is:
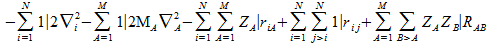
- ZA is the nuclear charge of the Ath atom
-
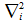 is the
Laplacian for the ith electron
is the
Laplacian for the ith electron
-
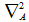 is the Laplacian for the nuclei
is the Laplacian for the nuclei
- RAB is the distance between the nuclei
- rij is the distance between the electrons
- riA the distance between an electron and a nucleus
Currently it is impossible to find an exact mathematical solution to such a differential equation. Scientists found an approximate solution for the molecular wave-function by using various physical and mathematical principles. In de sequel we explain what you need to understand in order to think up a model and to analyse the calculated results.
Copyright© 2023 Demeester Piet